Open SiteMaster (OSM)
L3 software that acts as a hub coordinating the actions of your plant, enabling all the management of serialization and aggregation processes
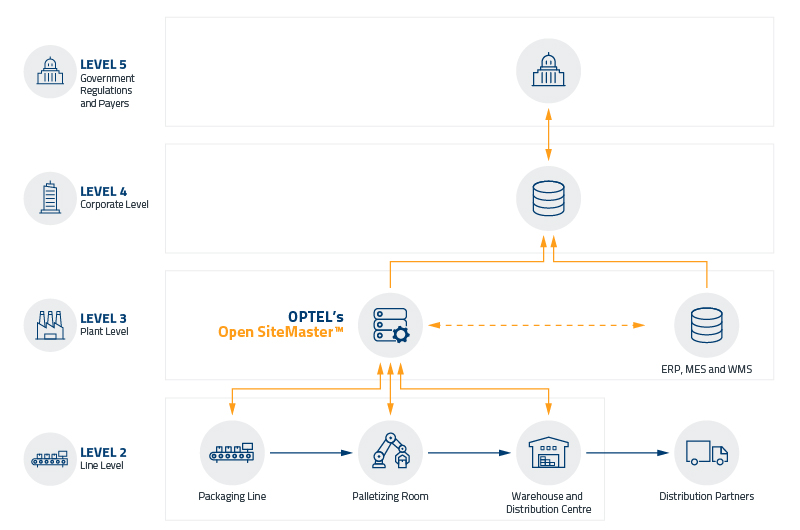

Allows web-based access from anywhere within the plant, to an unlimited number of users, controlling viewable content and availability of functions based on group roles and assigned access rights.

Adapts to your business logic and processes by customizing actions and triggers to fully configurable workflows.

Limits the number of exchanges with the corporate serialization server and changes in data ownership by keeping your production data at the plant level until all actions are completed (full-batch rework, QA sample, QA approval, etc.).

Complies with numerous laws and regulations around the world, and support industry standards and best practices.


Open SiteMaster (OSM)
L3 software that acts as a hub coordinating the actions of your plant, enabling all the management of serialization and aggregation processes

Kompano
Solution that manages the serialization of products in the warehouse: prints labels, reworks, sending and receiving

LineMaster
Software embedded in the line that records all serialization and aggregation events at the packaging level

Vision Systems
Discover OPTEL’s unmatched Vision Systems with AI capabilities. Our Visions Systems can inspect any product with high accuracy to ensure product safety and quality.

Serialization
Serialization is the first step in the traceability journey. OPTEL delivers scalable pharma solutions for global compliance and end-to-end visibility.

Aggregation
Aggregation takes you beyond unit-level traceability to see the whole picture

VerifyBrand™ for the Pharmaceutical Industry
VerifyBrand™ is an essential tool to comply with most of the world’s pharmaceutical regulations
