35
Years of Expertise
6000
Systems Installed Worldwide
25B
Products Tracked Per Year
24 / 7
International Tech Support
96 %
Satisfaction Level Of Service




































Discover our end-to-end traceability solutions
SUPPLY CHAIN INTELLIGENCE SOLUTION
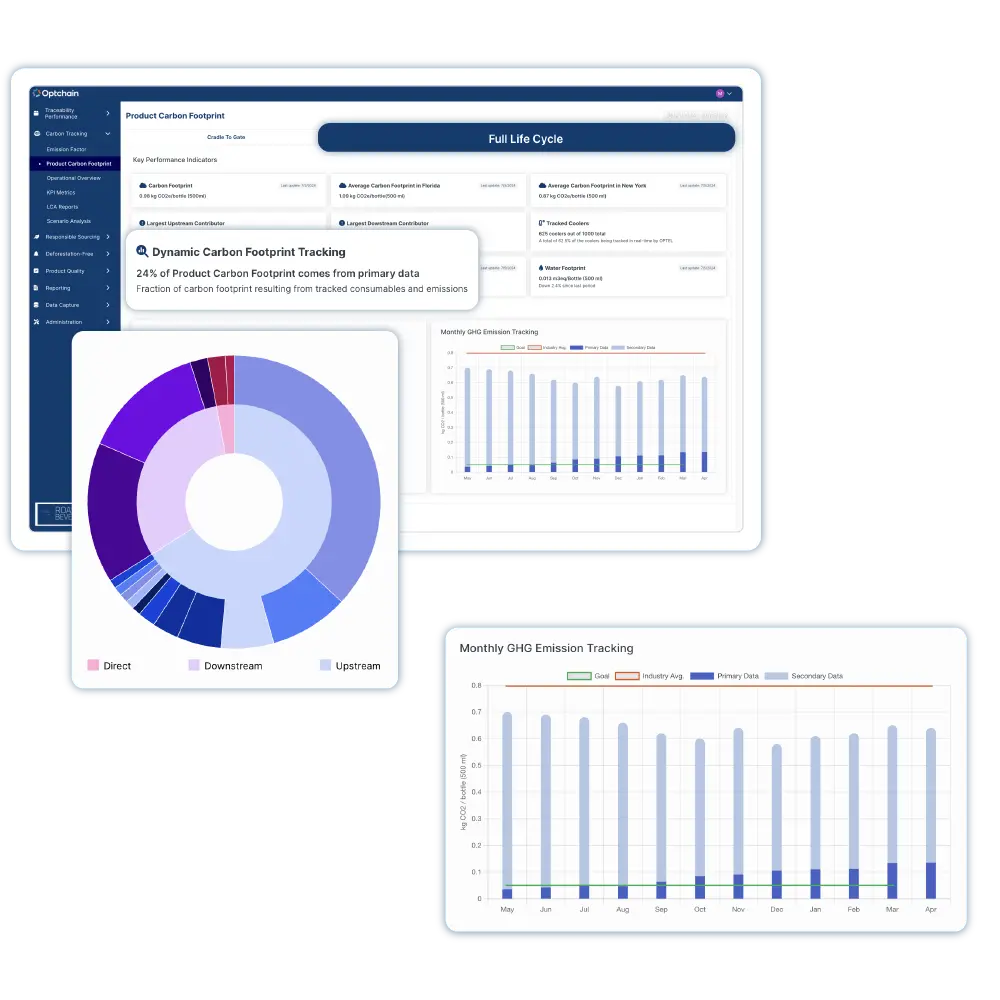
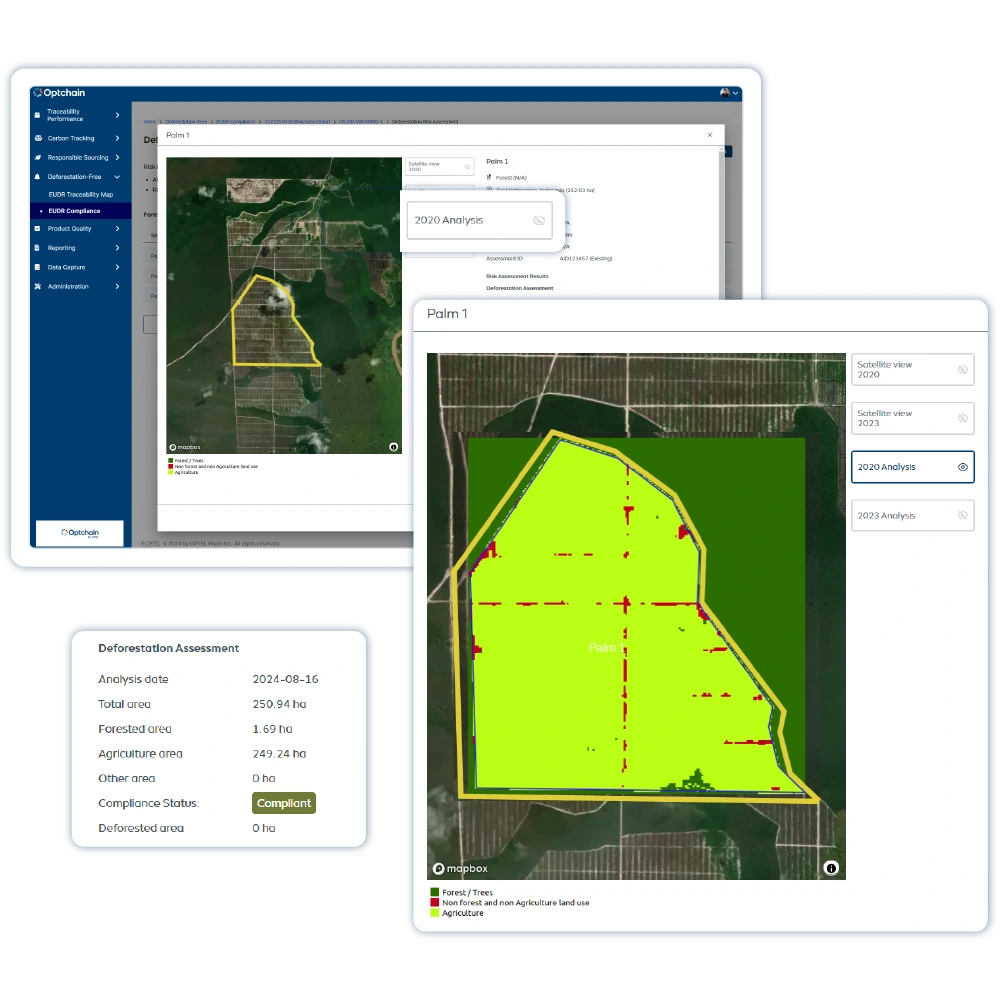
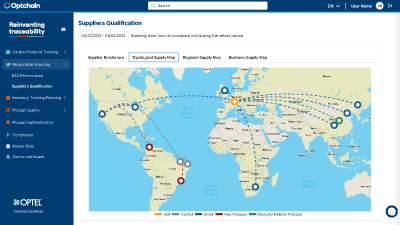
The Optchain platform delivers multi-tier visibility, enables ESG and product compliance, and empowers businesses to identify, prioritize, and mitigate upstream supply chain risks at scale.
Discover Optchain™

TRACK & TRACE AND INSPECTION SYSTEMS
Site-level serialization and aggregation hardware solutions to optimize line performance and ensure track-and-trace excellence with AI-integrated vision systems.
Discover TrackSafe
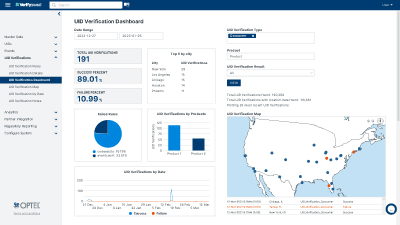
SUPPLY CHAIN INTEGRITY SOLUTION
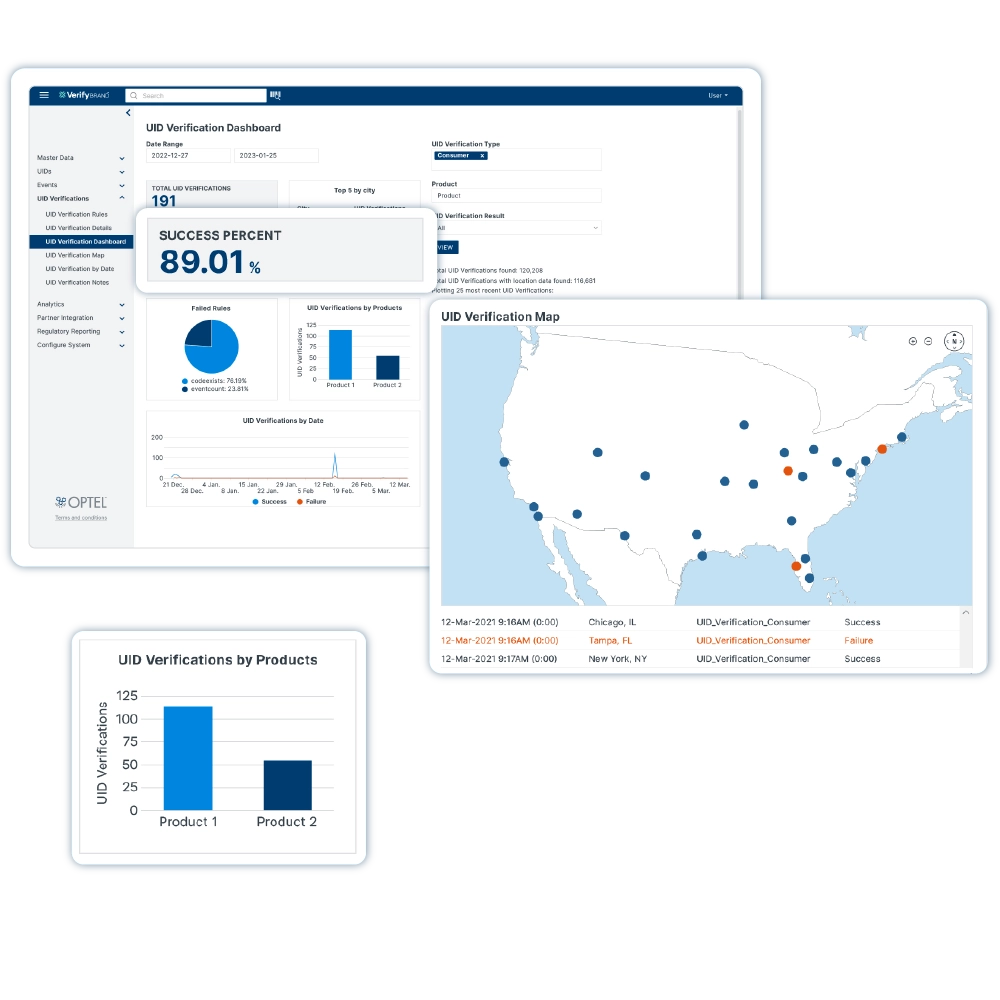
VerifyBrand™ is a real-time serialization and traceability software ensuring compliance, enhancing brand protection, authenticating products, and optimizing inventory.
Discover VerifyBrand™