TRACK . OPERATE . TRUST
For over two decades, OPTEL’s patented VerifyBrand™ solution – an enterprise-level serialization and traceability solution – has empowered leading global brands to track products, safeguard assets, and ensure regulatory compliance.
WHAT MAKES VERIFYBRAND™ SOFTWARE UNIQUE


EASY TO LEARN
- Onboard training included
- Upgrade support and documentation
- Technical support available anytime


EASY TO USE
- Intuitive user-interface
- Available in several languages
- Cloud based SaaS making it very flexible
- Automated workflow


EASY TO BUDGET
- Fixed-fee cost model
- Pricing structure transparent
- Free upgrades

EASY TO TRACK
- Track and trace module
- Regulation compliance guaranteed
- Partners insights to leverage actionable intelligence
- Leveraging the GS1 EPCIS standard

EASY TO MIGRATE
- Provides the right solution for data migration project
- Online data transfer and offline data transfer needs addressed
- Data is physically or logically segregated from other clients’data
- Scalable and secure (adheres to OWASP Secure Coding’s practices)
- Dedicated account management
- Turnkey partner integration


EASY TO DRIVE PERFORMANCE
- Single-tenant architecture
- Highly configurable with modules and adds-on
- Built-in and advanced analytics reporting
- Fast response for items batch history and serial numbers queries
- Execution against customer timelines
- No delayed deployments


FEATURES AND BENEFITS ABOUT VERIFYBRAND™ SOFTWARE
HAVE A GLIMPSE OF VERIFYBRAND™

Compliance with Global Standards
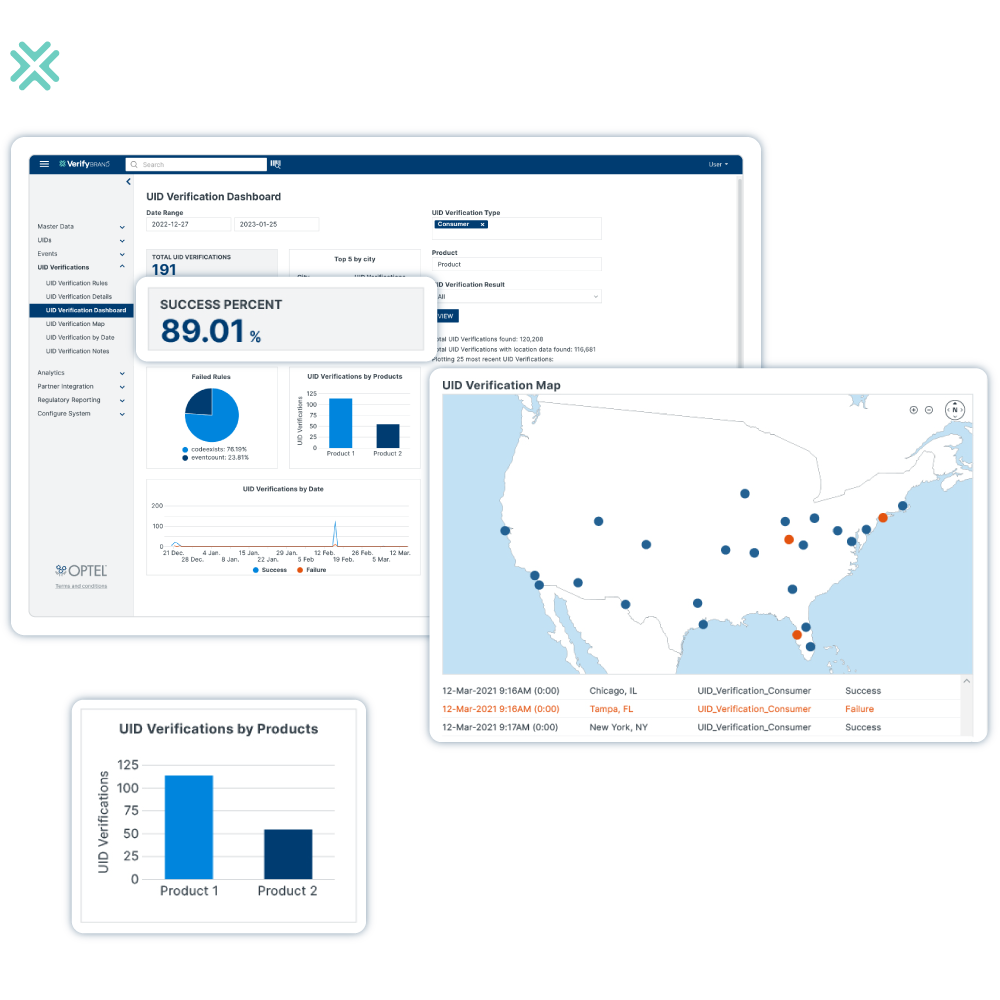
VerifyBrand™ streamlines regulatory compliance with a one-stop solution that connects your supply chain with government authorities. Our solution supports over 10 life sciences regulations (including US DSCSA, EU FMD) and US FSMA for Food & Beverage.
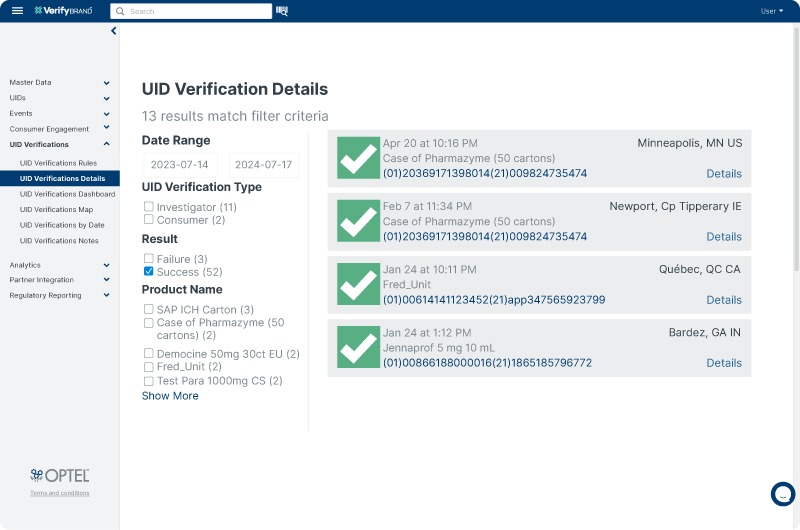

Product Authenticity Verification
VerifyBrand™ is the perfect tool to verify the authenticity of any product through your supply chain. It is an essential tool to combat counterfeiting or fraud by executing investigations in your operations or through consumer-related apps. With its high configurability, VerifyBrand™ can be tailored to meet your specific verification requirements, ensuring seamless integration into your existing processes.
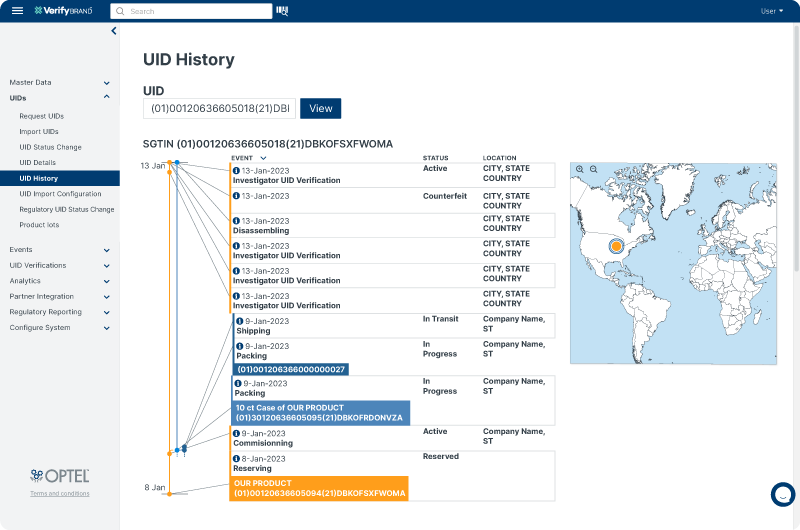

Product Lifecycle Monitoring
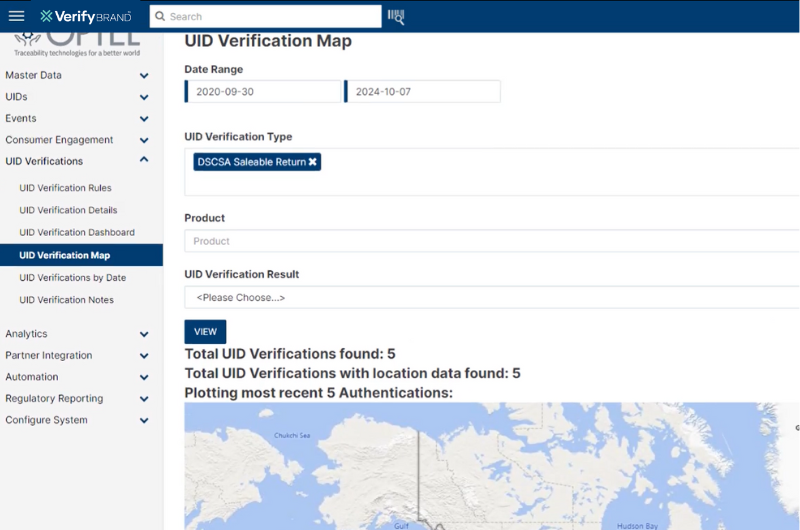
VerifyBrand™ provides a complete view of the product’s lifecycle by tracking key events throughout its journey. With easy-to-use geolocation features, it allows you to quickly pinpoint a product’s location and conduct deeper investigations when needed. The platform is user-friendly, offering intuitive map-based visualization for effortless tracking. Additionally, it helps enforce policies by ensuring products are sold or returned to the appropriate region. By leveraging trade data, the solution also supports the development of programs aimed at discouraging parallel trade.
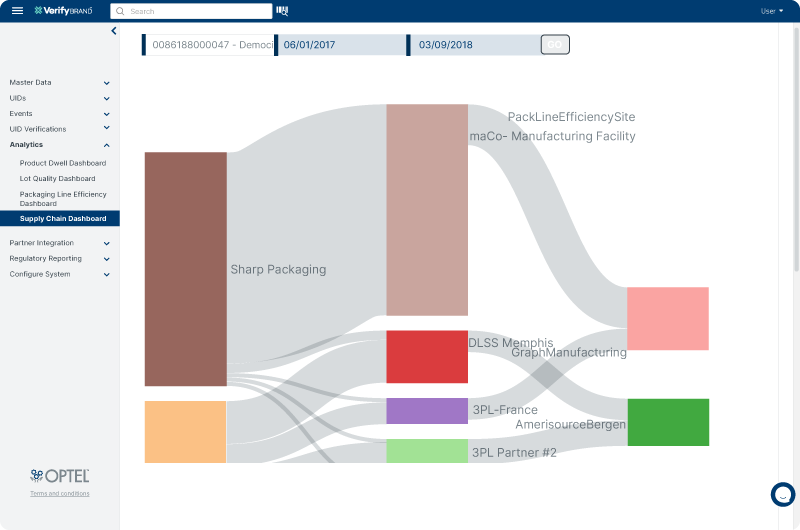

Real-time Visibility
VerifyBrand™ provides real-time visibility across your supply chain operations, allowing you to track multiple shipments sent to trading partners related to a specific lot. You can view the entire journey of the lot, including a breakdown of its status. It is also possible to see the quantity associated with each serial number unit in collaboration with your partners.
SIMPLE, SCALABLE TRACEABILITY FOR GROWING MANUFACTURERS
VerifyBrand™ FLEX is OPTEL’s streamlined track-and-trace solution designed for mid-sized manufacturers, part of the VerifyBrand™ portfolio.
It delivers powerful traceability—without complex systems, costly integrations, or bulky hardware.
FLEX adapts to your operations and grows with you.
With FLEX, you can:
- Prevent counterfeiting with product authentication
- Spot diversion and unauthorized distribution
- Gain real-time visibility across your channels
- Simplify recalls and batch-level tracking
- Build trust with partners and end-users


How TruPharma Gained Control of DSCSA Compliance
Case StudyVERIFYBRAND™ CAN HELP YOU WITH


QR Code Verification
OPTEL’s QR Code Verification solution enables accurate authentication of any product through a cloud platform. It allows customers to verify the authenticity of the scanned product.
Learn more


Reusable Asset Management
OPTEL’s Reusable Asset Management (RAM) solution allows manufacturers to track their reusable packaging from manufacturing sites to end user and back.
Learn more
35
Years of Expertise
6000
Systems Installed Worldwide
25B
Products Tracked Per Year
24 / 7
International Tech Support
96 %
Satisfaction Level Of Service