API Traceability
The Ministry of Health and Family Welfare (Govt. of India) on January 18, 2022, has issued the Drugs (Amendment) Rules, 2022 to further amend the Drugs Rules, 1945.
The amendment makes QR codes mandatory on every active pharmaceutical ingredient (API). It states that every API imported or manufactured in India shall bear a QR (Quick Response) code on the label and at every packaging level. The QR code must encase essential data that can be read through computer software for efficient tracking and tracing.
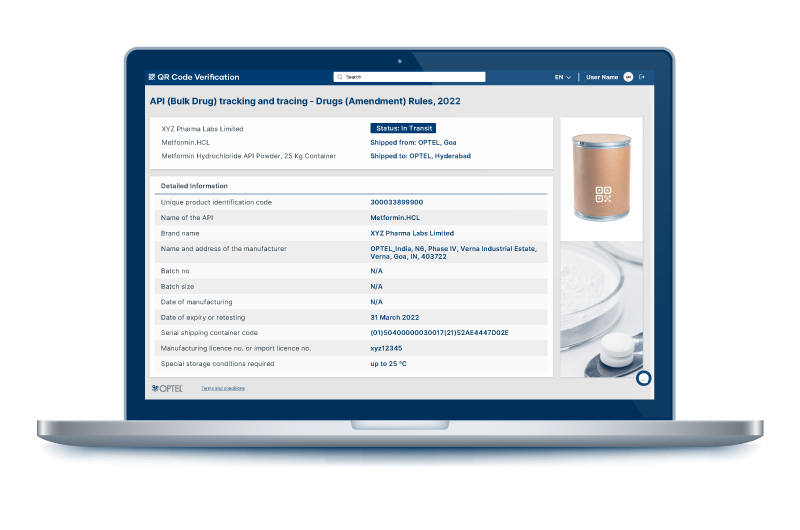
IT MUST CONTAIN THESE 11 DATA POINTS:
- Unique product identification code;
- Name of the API;
- Brand name (if any);
- Name and address of the manufacturer;
- Batch no.;
- Batch size;
- Date of manufacturing;
- Date of expiry or retesting;
- Serial shipping container code (SSCC);
- Manufacturing licence no. or import licence no.;
- Special storage conditions required (if any).
The QR code has been made mandatory for the top 300 widely used drug brands produced from August 1st 2023 — as part of the Centre’s ‘track and trace’ mechanism to check the sale of counterfeit and spurious medicines.
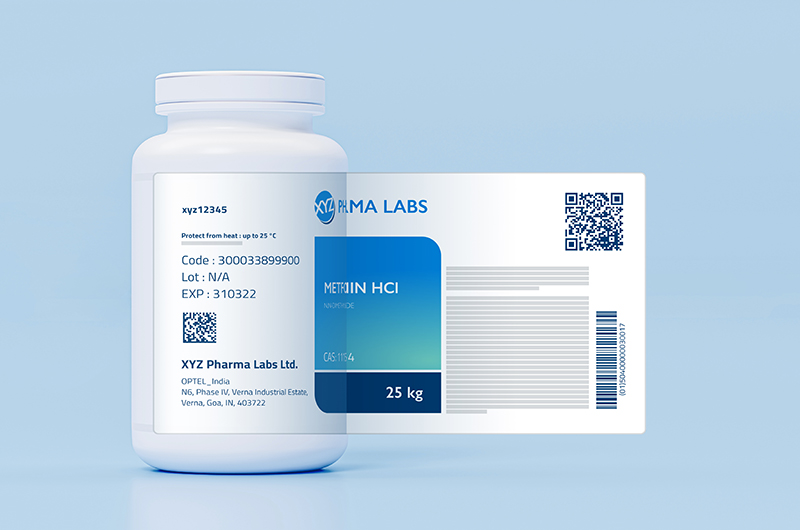
TOP 300 DRUGS TO HAVE QR CODES FROM AUGUST 1ST, 2023
Also, in June 2022, the Department of Health and Family Welfare has issued a notification that, from August 1st, 2023, the top 300 widely used drug brands must have QR codes printed on their primary packaging or in their secondary packaging if not enough space in the primary package label.
They need to store the following data or information:
- Unique Product Identification Code
- Proper and Generic Name of the drug
- Brand Name
- Name and address of the manufacturer
- Batch number
- Date of Manufacturing
- Date of Expiry
- Manufacturing Licence Number
This is part of the Centre’s ‘track and trace’ mechanism to check the sale of counterfeit and spurious medicines.

OUR SOLUTION:
Powered by VerifyBrand™ our downstream serialization traceability software, OPTEL’s QR Code Verification solution enables customers to meet the regulation requirements.
The Ministry of Health and Family Welfare published guidelines for barcoding the Top 300 drug brands in order to combat counterfeiting, diversion, and unauthorized sales.
QR codes need to be printed on or affixed to the primary packaging (if there is no space on the primary package the codes could be placed on the secondary packaging).
OPTEL’s QR Code Verification solution enables accurate authentication of any product through a cloud platform. It allows customers to verify the authenticity of the scanned product.

FLYER QR CODE Verification
KEY BENEFITS
- A cloud platform that offers exceptional flexibility to meet high demand for QR code scans
- An user-friendly and readable interface
- 100% accurate authentication
- Could be used for non-serialized items as well
- Effortless and exact verification and authentication of any product
- Can be easily integrated with other OPTEL’s solutions.
INTERESTED IN OUR SOLUTION?
Related Solutions

VerifyBrand™ for the Pharmaceutical Industry
VerifyBrand™ is a serialization software helping pharmaceutical manufacturers to comply with traceability regulations.

Vision Systems
Ensure pharmaceutical compliance and product quality with OPTEL’s AI-powered vision systems. Achieve 100% inspection and seamless integration.

Serialization
Serialization is the first step in the traceability journey. OPTEL delivers scalable pharma solutions for global compliance and end-to-end visibility.
RELATED POST

How to Reach End-to-End Visibility in Pharma Supply Chains
End-to end visibility in pharma supply chains: Discover the significance of supply chain visibility and the tactics for its implementation.

Tackling the Pharmaceutical Industry’s Labour Shortage Head-on with Automation
Solve the labour shortage dilemma by embracing automation. Learn more

Overview of the Major Pharmaceutical Regulations
Explore the major pharmaceutical regulations in the world and let us guide you through this compliance journey

Drugs (Amendment) Rules, 2022 For Active Pharmaceutical Ingredient (Bulk Drug) Manufactured or Imported in India
The amendment makes QR codes mandatory on every active pharmaceutical ingredient. The Amendment Rules will come into force from January 01, 2023.
