What Are Vision Systems in Pharmaceutical Manufacturing?
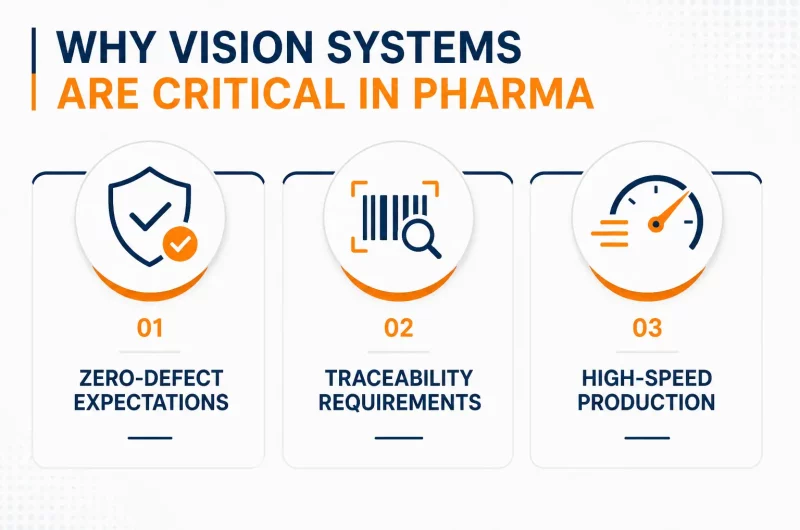
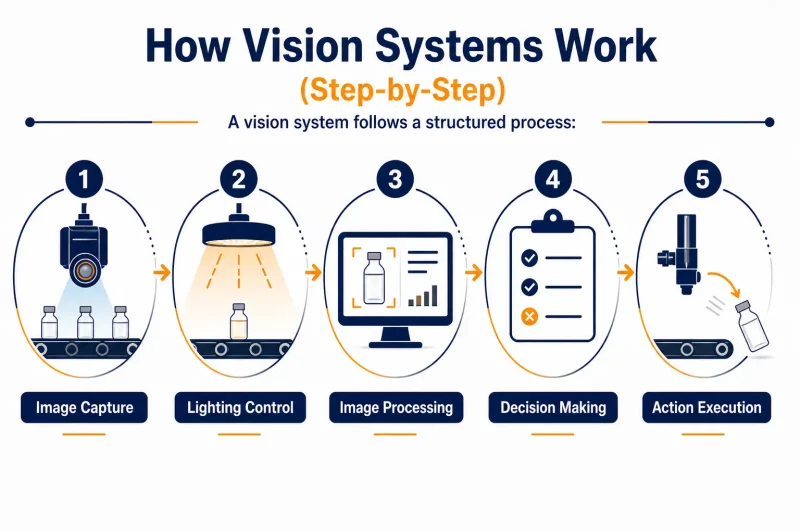
Vision systems are automated inspection technologies that use cameras, lighting, and software to analyze products on production lines. Their role is simple: detect defects, verify compliance, and ensure consistency at high speed.
In pharma, this includes tasks like checking tablet integrity, verifying label accuracy, and confirming serialization codes. Unlike manual inspection, vision systems operate continuously and with repeatable accuracy.
For example, on a blister packaging line, a vision system can instantly detect a missing tablet or a broken seal, something a human operator might miss after hours of repetitive work.