35
Anos de Experiência
6000
Sistemas Instalados em Todo o Mundo
25B
Bilhões de Produtos Rastreados por Ano
24 / 7
Suporte Técnico Internacional
96 %
Nível de Satisfação de






































DESCUBRA NOSSA SOLUÇÃO DE RASTREABILIDADE DE PONTA A PONTA
PLATAFORMA DE INTELIGÊNCIA DA CADEIA DE SUPRIMENTOS
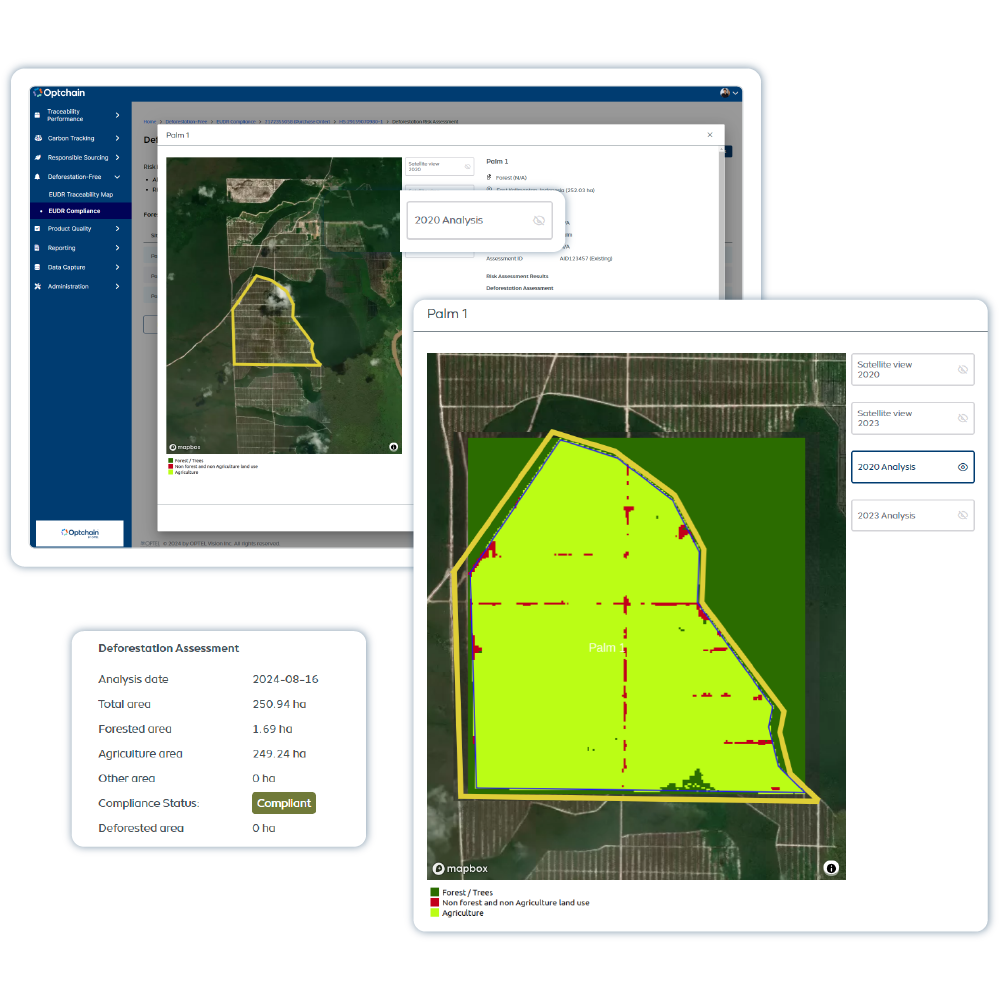
A solução Optchain oferece visibilidade em múltiplos níveis, viabiliza a conformidade com requisitos ESG e regulatórios de produtos e permite que as empresas identifiquem, priorizem e mitiguem riscos a montante da cadeia de suprimentos em escala.
Descubra o Optchain

Sistemas de Rastreamento e Inspeção
Soluções hardware de serialização e agregação para otimizar o desempenho da linha e garantir excelência em track & trace com sistemas de visão integrados à IA.
Descubra o TrackSafe
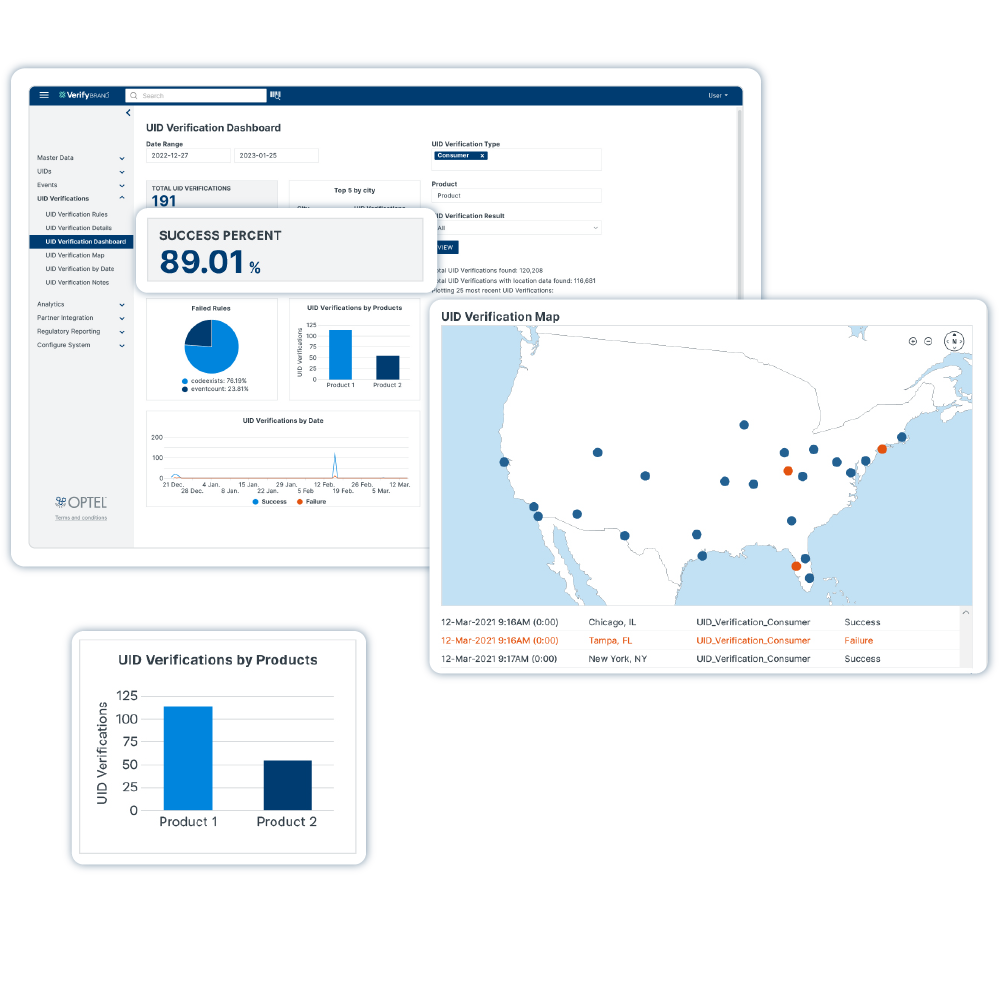
Solução de Integridade da Cadeia de Suprimentos
VerifyBrand™ é um software de serialização e rastreabilidade real que garante conformidade com regulamentações, aprimora a proteção da marca, autentica produtos e otimiza o estoque.
Descubra o VerifyBrand™